 Since four orbitals are involved it may undergo sp 3 hybridisation. On substituting this values to the above equation then, Cationic and anionic charge is 0 and 1 respectively. There is no monovalent atom present here. Here the valence electrons in chlorine is 7. Hybridisation = ½[ valence electrons – monovalent atom – cationic The hybridisation in any ionic molecule can be found through the following equation. Shape and Bond Angle of ClO 3 – Hybridisation in Chlorate anion, ClO 3 – The bond length of Cl-O bonds are same and is 1.49 x 10 -10m. To reduce this factor the atoms will stay as far as apart to form triagonal pyramidal geometry with bond angle 107 0. There is strong repelsion between the lone pairs in chlorine and oxygen. 
But the presence of lone pair in chlorine atoms don’t allows this geometry. The sp 3 hybridisation followed by chlorate ion has to be in tetrahedral in shape. That is A is central atom(Cl), X is atom attached into central atom(Oxygen), N is the number of lone pairs in central atom(1). Shape and angle of ClO 3 –Īccording to VSEPR theory atoms occupies a position around the central atom in such a way to reduce repelsion between the electrons associated with atoms. 
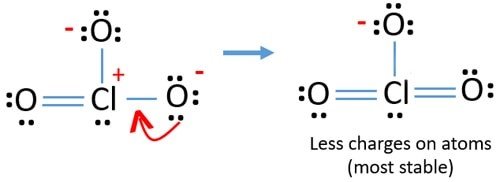
The lone pairs present in chlorate anion is 8 pairs.Ĭhlorine contains one pair. Lone pair of electrons in Chlorate anion,ClO 3 – The total number of electrons involved in bond making of chlorate anion is 26. The total number of electrons involved in bond making process is called the valence electrons. Valence electrons in Chlorate anion, ClO 3 – The formal charge of atoms in chlorate anion is zero for two chlorine and two oxygen atoms. of bondsįormal charge in one oxygen with single bond = 6-6-1 = -1 Distribution should be done in such a way that the atoms obey octet rule.įormal charge = valence electrons – No. To make the bond there took six electrons so the remaining 20 electrons are distributed equally in all atoms. Form one bond between chlorine and oxygen.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
